
Proprioception!
Wilkinson Lab Goals
1) To understand how the muscle spindle senses muscle stretch and how the sensitivity of muscle spindle afferents are controlled.
2) To understand how environmental conditions and/or disease states impair the function of the muscle spindle.
Main Experimental Techniques
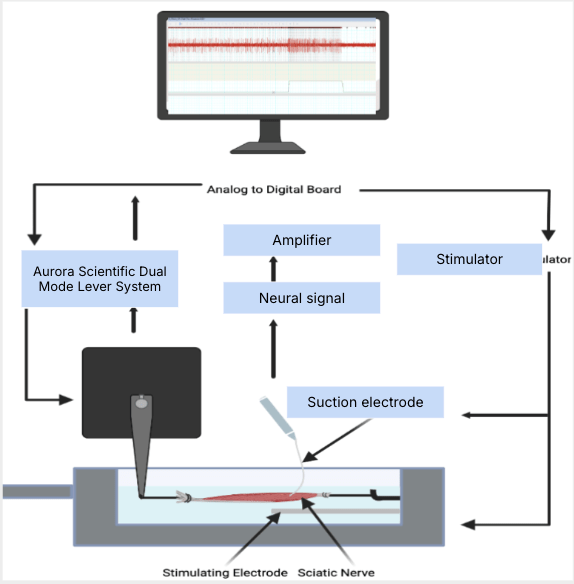
Electrophysiology
- Involves muscle extraction and collecting neural data through stretching the muscle (EDL) to 3 different lengths and recording any neural activity sent through the sciatic nerve
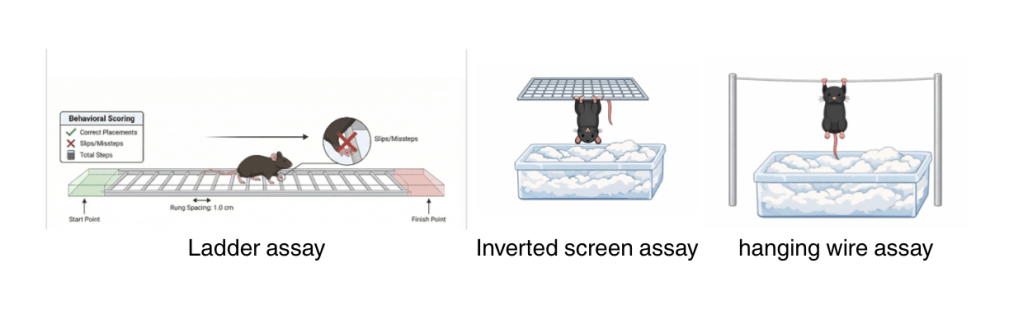
Behavior Assays
- Ladder walking assay; the animal model walks across a series of rungs, and any missteps or gait issues are noted
- Inverted screen assay; the animal model is placed on a screen which is inverted for two minutes. The time it takes for the animal to fall is noted.
- Wire hanging assay; the animal model is suspended on a wire, and the respective drop times are noted.
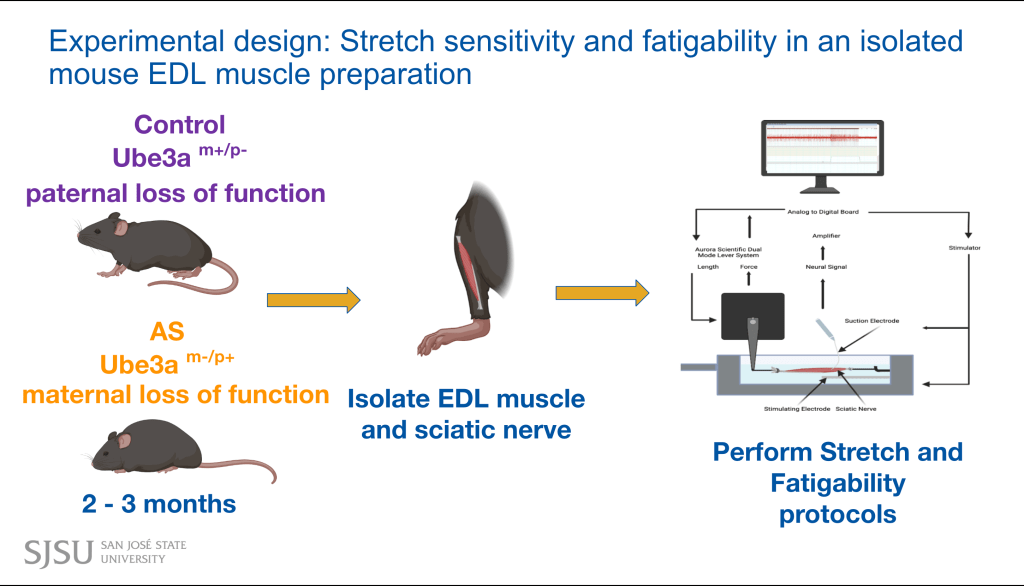
Current Research
- Muscle proprioceptor function and behavior comparisons in Angelman Syndrome (AS) mouse models
- Behavior assays are done to examine any differences in gait/slipups/drop times between mice with or without the maternal loss of Ube3a.
- Electrophysiology is done with the EDL and sciatic nerve of mice with the paternal (control) or maternal (AS experiment) loss of Ube3a. Any exhibited neural data comparisons are noted.
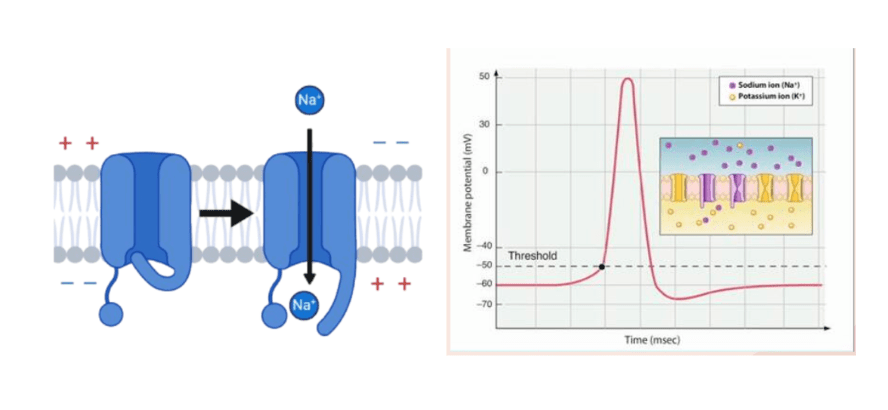
- The role of voltage gated sodium channel subtypes in muscle spindle afferent (MSA) function
- Muscle spindle afferent functions enable proprioceptive function, and different voltage gated sodium (Nav) channels are involved. We are interested in examining the different functions these Nav channels play in MSA function, and whether they may differ by age. Specific channels of interest are Nav1.6, Nav1.1, and Nav1.2.
- The impact of chemotherapy drugs on behavioral motor function and CIPN (chemotherapy induced peripheral neuropathy)
- CIPN is peripheral nerve damage that results as a side effect from chemotherapy treatments and can impact motor and sensory functions.
- We examine the gait and drop time differences in animal models with or without chemotherapy drugs (saline vs. PTX) through the ladder and inverted screen assays.
- Immunohistochemistry is then performed on the paws to note any nerve damage.
Past Research
- Control of gamma motor neuron activity using optogenetics
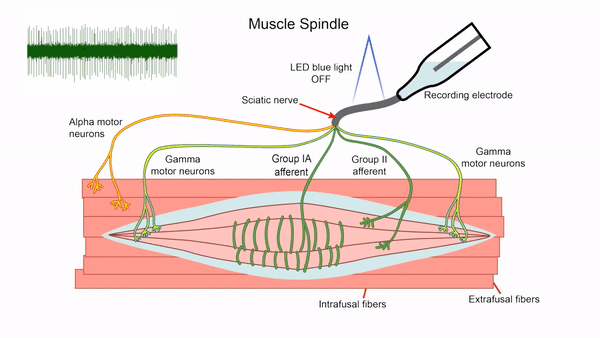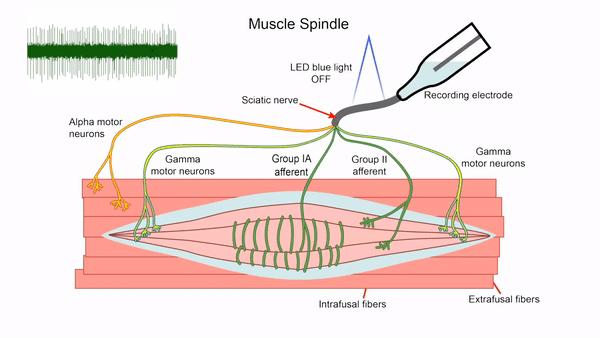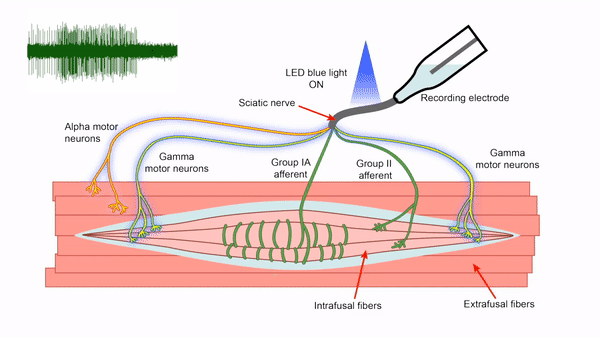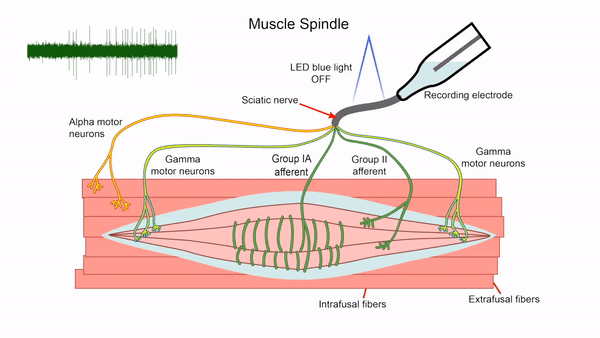
We are currently developing an optogenetics technique to study the gamma motor neurons that innervate the muscle spindle and are vital for motor control. The length of the muscle spindle is controlled by gamma motor neurons innervating the intrafusal muscle fibers of the spindle. However, it has been challenging to study the gamma motor neurons since it is hard to specifically stimulate the small gamma but not the larger alpha motor neurons that control the force generating extrafusal fibers using electrical stimulation. We express the blue light activated channelrhodopsin2 (ChR2) in ChAT positive motor neurons and use lower optical intensities of light to recruit the small gamma motor neurons first. We plan to use this technique to study gamma motor neuron function in vitro. This tool will also allow us to assay intrafusal fiber function in disease states.
- Modulation of muscle spindle afferent excitability by glutamate
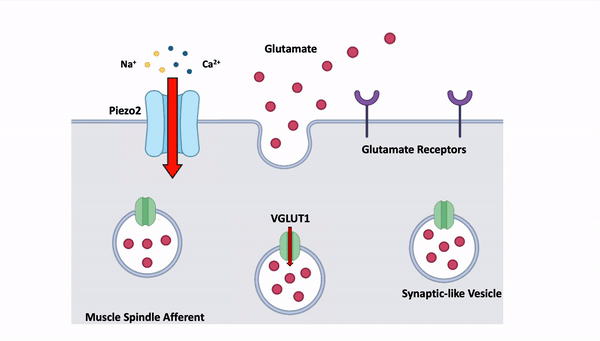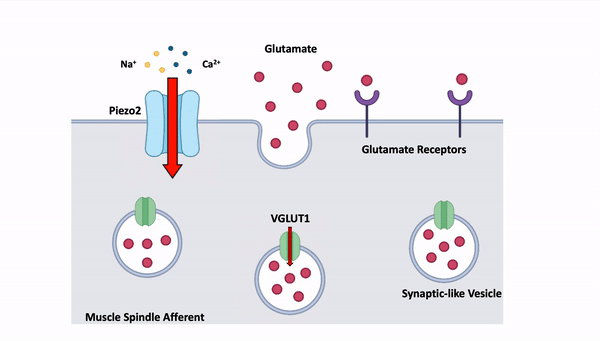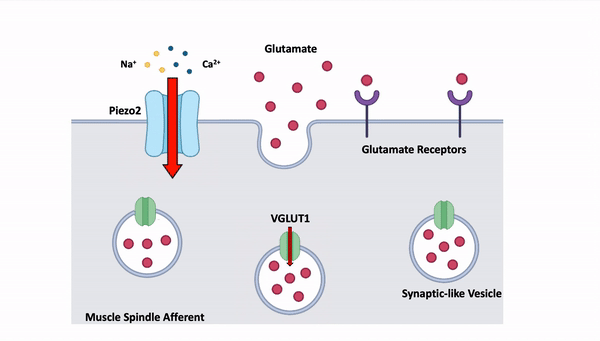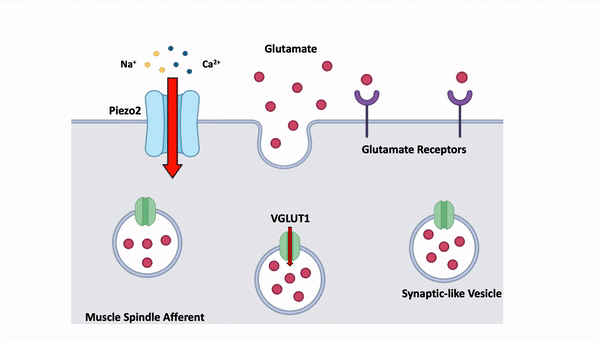
Synaptic-like vesicles containing glutamate are found within muscle spindle afferent peripheral nerve endings. These vesicles are released in an activity dependent manner, with an increase in vesicle release following multiple stretches or high frequency vibration. This glutamate is thought to increase afferent sensitivity to stretch as exogenous glutamate can increase whole nerve firing rate during stretch. However, the effect on individual afferents has not been determined. We are characterizing the precise role of glutamate in the functioning of muscle spindle afferents, the sensory neurons responsible for proprioception or the ability to sense one’s body position. Currently, we are directly recording afferent activity and stretch response in muscles from both wild-type mice and those lacking a copy of the vesicular glutamate transporter 1 (VGLUT1) gene. Since the VGLUT1 gene encodes for a protein needed for glutamate packaging and release, we expect to see altered or aberrant stretch response in the afferents of mice that lack a single copy of this gene. Through direct recordings we hope to deepen our understanding of the role of glutamate in proprioception.